Contacts & Chemistry
If you would like to know more about neuropsychology, you can contact me here: http://answers.yahoo.com/my-activity
THE PERIODIC TABLE
There are 18 vertical columns, or groups, in the standard periodic table. At present, there are three versions of the periodic table, each with its own unique column headings, in wide use. The three formats are the old International Union of Pure and Applied Chemistry (IUPAC) table, the Chemical Abstract Service (CAS) table, and the new IUPAC table. The old IUPAC system labeled columns with Roman numerals followed by either the letter A or B. Columns 1 through 7 were numbered IA through VIIA, columns 8 through 10 were labeled VIIIA, columns 11 through 17 were numbered IB through VIIB and column 18 was numbered VIII. The CAS system also used Roman numerals followed by an A or B. This method, however, labeled columns 1 and 2 as IA and IIA, columns 3 through 7 as IIIB through VIB, column 8 through 10 as VIII, columns 11 and 12 as IB and IIB and columns 13 through 18 as IIIA through VIIIA. However, in the old IUPAC system the letters A and B were designated to the left and right part of the table, while in the CAS system the letters A and B were designated to the main group elements and transition elements respectively. (The preparer of the table arbitrarily could use either an upper-or lower-case letter A or B, adding to the confusion.) Further, the old IUPAC system was more frequently used in Europe while the CAS system was most common in America. In the new IUPAC system, columns are numbered with Arabic numerals from 1 to 18. These group numbers correspond to the number of s, p, and d orbital electrons added since the last noble gas element (in column 18). This is in keeping with current interpretations of the periodic law which holds that the elements in a group have similar configurations of the outermost electron shells of their atoms. Since most chemical properties result from outer electron interactions, this tends to explain why elements in the same group exhibit similar physical and chemical properties. Unfortunately, the system fails for the elements in the first 3 periods (or rows; see below). For example, aluminum, in the column numbered 13, has only 3 s, p, and d orbital electrons. Nevertheless, the American Chemical Society has adopted the new IUPAC system.
The horizontal rows of the table are called periods. The elements of a period are characterized by the fact that they have the same number of electron shells; the number of electrons in these shells, which equals the element's atomic number, increases from left to right within each period. In each period the lighter metals appear on the left, the heavier metals in the center, and the nonmetals on the right. Elements on the borderline between metals and nonmetals are called metalloids.
Group 1 (with one valence electron) and Group 2 (with two valence electrons) are called the alkali metals and the alkaline-earth metals, respectively. Two series of elements branch off from Group 3, which contains the transition elements, or transition metals; elements 57 to 71 are called the lanthanide series, or rare earths, and elements 89 to 103 are called the actinide series, or radioactive rare earths; a third set, the superactinide series (elements 122-153), is predicted to fall outside the main body of the table, but none of these has yet been synthesized or isolated. The nonmetals in Group 17 (with seven valence electrons) are called the halogens. The elements grouped in the final column (Group 18) have no valence electrons and are called the inert gases, or noble gases, because they react chemically only with extreme difficulty.
In a relatively simple type of periodic table, each position gives the name and chemical symbol for the element assigned to that position; its atomic number; its atomic weight (the weighted average of the masses of its stable isotopes, based on a scale in which carbon-12 has a mass of 12); and its electron configuration, i.e., the distribution of its electrons by shells. The only exceptions are the positions of elements 103 through 118; complete information on these elements has not been compiled. Larger and more complicated periodic tables may also include the following information for each element: atomic diameter or radius; common valence numbers or oxidation states; melting point; boiling point; density; specific heat; Young's modulus; the quantum states of its valence electrons; type of crystal form; stable and radioactive isotopes; and type of magnetism exhibited by the element (paramagnetism or diamagnetism).
Organized array of all the chemical elements in approximately increasing order of their atomic weight. The elements show a periodic recurrence of certain properties, first discovered in 1869 by Dmitry I. Mendeleyev. Those in the same column (group) of the table as usually arranged have similar properties. In the 20th century, when the structure of atoms was understood, the table was seen to precisely reflect increasing order of atomic number. Members of the same group in the table have the same number of electrons in the outermost shells of their atoms and form bonds of the same type, usually with the same valence; the noble gases, with full outer shells, generally do not form bonds. The periodic table has thus greatly deepened understanding of bonding and chemical behaviour. It also allowed the prediction of new elements, many of which were later discovered or synthesized. For an illustration of the periodic table, see chemical element.
Some versions of the table show a dark stair-step line along the metalloids. Metals are to the left of the line and non-metals to the right.
Arrangement
The layout of the periodic table demonstrates recurring ("periodic") chemical properties. Elements are listed in order of increasing atomic number (i.e. the number of protons in the atomic nucleus). Rows are arranged so that elements with similar properties fall into the same vertical columns ( "groups" ). According to quantum mechanical theories of electron configuration within atoms, each horizontal row ( "period" ) in the table corresponded to the filling of a quantum shell of electrons. There are progressively longer periods further down the table, grouping the elements into s- , p- , d- and f-blocks to reflect their electron configuration.
In printed tables, each element is usually listed with its element symbol and atomic number; many versions of the table also list the element's atomic mass and other information, such as its abbreviated electron configuration, electronegativity and most common valence numbers.
As of 2006, the table contains 117 chemical elements whose discoveries have been confirmed. Ninety-four are found naturally on Earth, and the rest are synthetic elements that have been produced artificially in particle accelerators. Elements 43 (technetium), 61 (promethium), 93 (neptunium) and 94 (plutonium) have no stable isotopes and were first discovered synthetically; however, they were later discovered in trace amounts on earth as products of natural radioactive decay processes.
Periodicity of chemical properties
The main value of the periodic table is the ability to predict the chemical properties of an element based on its location on the table. It should be noted that the properties vary differently when moving vertically along the columns of the table than when moving horizontally along the rows.
Groups and periods
A group is a vertical column in the periodic table of the elements.
Groups are considered the most important method of classifying the elements. In some groups, the elements have very similar properties and exhibit a clear trend in properties down the group — these groups tend to be given trivial (unsystematic) names, e.g. the alkali metals, alkaline earth metals, halogens and noble gases. Some other groups in the periodic table display fewer similarities and/or vertical trends (for example Groups 14 and 15), and these have no trivial names and are referred to simply by their group numbers.
A period is a horizontal row in the periodic table of the elements.
Although groups are the most common way of classifying elements, there are some regions of the periodic table where the horizontal trends and similarities in properties are more significant than vertical group trends. This can be true in the d-block (or " transition metals"), and especially for the f-block, where the lanthanides and actinides form two substantial horizontal series of elements.
Periodic trends of groups
Modern quantum mechanical theories of atomic structure explain group trends by proposing that elements within the same group have the same electron configurations in their valence shell, which is the most important factor in accounting for their similar properties. Elements in the same group also show patterns in their atomic radius, ionization energy, and electronegativity. From top to bottom in a group, the atomic radii of the elements increase. Since there are more filled energy levels, valence electrons are found farther from the nucleus. From the top, each successive element has a lower ionization energy because it is easier to remove an electron since the atoms are less tightly bound. Similarly, a group will also see a top to bottom decrease in electronegativity due to an increasing distance between valence electrons and the nucleus.
Periodic trends of periods
Elements in the same period show trends in atomic radius, ionization energy, electron affinity, and electronegativity. Moving left to right across a period, atomic radius usually decreases. This occurs because each successive element has an added proton and electron which causes the electron to be drawn closer to the nucleus. This decrease in atomic radius also causes the ionization energy to increase when moving from left to right across a period. The more tightly bound an element is, the more energy is required to remove an electron. Similarly, electronegativity will increase in the same manner as ionization energy because of the amount of pull that is exerted on the electrons by the nucleus. Electron affinity also shows a slight trend across a period. Metals (left side of a period) generally have a lower electron affinity than nonmetals (right side of a period) with the exception of the noble gases.
Examples
Noble gases
All the elements of Group 18, the noble gases, have full valence shells. This means they do not need to react with other elements to attain a full shell, and are therefore much less reactive than other groups. Neon followed by helium are the most inert elements among noble gases, since reactivity, in this group, increases with the periods: it is possible to make heavy noble gases react since they have much larger electron shells. However, their reactivity remains very low in absolute terms.
Halogens
In Group 17, known as the halogens, elements are missing just one electron each to fill their shells. Therefore, in chemical reactions they tend to acquire electrons (the tendency to acquire electrons is called electronegativity). This property is most evident for fluorine (the most electronegative element of the whole table), and it diminishes with increasing period.
As a result, all halogens form acids with hydrogen, such as hydrofluoric acid, hydrochloric acid, hydrobromic acid and hydroiodic acid, all in the form HX . Their acidity increases with higher period, for example, with regard to iodine and fluorine, since a large I − ion is more stable in solution than a small F − , there is less volume in which to disperse the charge.
Transition metals
For the transition metals (Groups 3 to 12), horizontal trends across periods are often important as well as vertical trends down groups; the differences between groups adjacent are usually not dramatic. Transition metal reactions often involve coordinated species.
Lanthanides and actinides
The chemical properties of the lanthanides (elements 57–71) and the actinides (elements 89–103) are even more similar to each other than the transition metals, and separating a mixture of these can be very difficult. This is important in the chemical purification of uranium concerning nuclear power.
Structure of the periodic table
The primary determinant of an element's chemical properties is its electron configuration, particularly the valence shell electrons. For instance, any atoms with four valence electrons occupying p orbitals will exhibit some similarity. The type of orbital in which the atom's outermost electrons reside determines the "block" to which it belongs. The number of valence shell electrons determines the family, or group, to which the element belongs.
The total number of electron shells an atom has determines the period to which it belongs. Each shell is divided into different subshells, which as atomic number increases are filled in roughly this order (the Aufbau principle):
Note that as atomic number (i.e. charge on the atomic nucleus) increases, this leads to greater spin-orbit coupling between the nucleus and the electrons, reducing the validity of the quantum mechanical orbital approximation model, which considers each atomic orbital as a separate entity.
Because of the importance of the outermost shell, the different regions of the periodic table are sometimes referred to as periodic table blocks, named according to the sub-shell in which the "last" electron resides, e.g. the s -block, the p -block, the d -block, etc.
The elements Ununbium, ununtrium, ununquadium, etc. are elements that have been discovered, but so far have not been named.
In Ancient Greece, the influential Greek philosopher Aristotle proposed that there were four main elements: air, fire, earth and water. All of these elements could be reacted to create another one; e.g. , earth and fire combined to form lava. However, this theory was dismissed when the real chemical elements started being discovered. Scientists needed an easily accessible, well organized database with which information about the elements could be recorded and accessed. This was to be known as the periodic table.
The original table was created before the discovery of subatomic particles or the formulation of current quantum mechanical theories of atomic structure. If one orders the elements by atomic mass, and then plots certain other properties against atomic mass, one sees an undulation or periodicity to these properties as a function of atomic mass. The first to recognize these regularities was the German chemist Johann Wolfgang Döbereiner who, in 1829, noticed a number of triads of similar elements.
In 1829 Döbereiner proposed the Law of Triads: The middle element in the triad had atomic weight that was the average of the other two members. The densities of some triads followed a similar pattern. Soon other scientists found chemical relationships extended beyond triads. Fluorine was added to Cl/Br/I group; sulfur, oxygen, selenium and tellurium were grouped into a family; nitrogen, phosphorus, arsenic, antimony, and bismuth were classified as another group.
This was followed by the English chemist John Newlands, who noticed in 1865 that when placed in order of increasing atomic weight, elements of similar physical and chemical properties recurred at intervals of eight, which he likened to the octaves of music, though his law of octaves was ridiculed by his contemporaries. However, while successful for some elements, Newlands' law of octaves failed for two reasons:
It was not valid for elements that had atomic masses higher than Ca.
When further elements were discovered, such as the noble gases (He, Ne, Ar), they could not be accommodated in his table.
Finally, in 1869 the Russian chemistry professor Dmitri Ivanovich Mendeleev and four months later the German Julius Lothar Meyer independently developed the first periodic table, arranging the elements by mass. However, Mendeleev plotted a few elements out of strict mass sequence in order to make a better match to the properties of their neighbors in the table, corrected mistakes in the values of several atomic masses, and predicted the existence and properties of a few new elements in the empty cells of his table. Mendeleev was later vindicated by the discovery of the electronic structure of the elements in the late 19th and early 20th century.
Earlier attempts to list the elements to show the relationships between them (for example by Newlands) had usually involved putting them in order of atomic mass. Mendeleev's key insight in devising the periodic table was to lay out the elements to illustrate recurring ("periodic") chemical properties (even if this meant some of them were not in mass order), and to leave gaps for "missing" elements. Mendeleev used his table to predict the properties of these "missing elements", and many of them were indeed discovered and fit the predictions well.
There are 18 vertical columns, or groups, in the standard periodic table. At present, there are three versions of the periodic table, each with its own unique column headings, in wide use. The three formats are the old International Union of Pure and Applied Chemistry (IUPAC) table, the Chemical Abstract Service (CAS) table, and the new IUPAC table. The old IUPAC system labeled columns with Roman numerals followed by either the letter A or B. Columns 1 through 7 were numbered IA through VIIA, columns 8 through 10 were labeled VIIIA, columns 11 through 17 were numbered IB through VIIB and column 18 was numbered VIII. The CAS system also used Roman numerals followed by an A or B. This method, however, labeled columns 1 and 2 as IA and IIA, columns 3 through 7 as IIIB through VIB, column 8 through 10 as VIII, columns 11 and 12 as IB and IIB and columns 13 through 18 as IIIA through VIIIA. However, in the old IUPAC system the letters A and B were designated to the left and right part of the table, while in the CAS system the letters A and B were designated to the main group elements and transition elements respectively. (The preparer of the table arbitrarily could use either an upper-or lower-case letter A or B, adding to the confusion.) Further, the old IUPAC system was more frequently used in Europe while the CAS system was most common in America. In the new IUPAC system, columns are numbered with Arabic numerals from 1 to 18. These group numbers correspond to the number of s, p, and d orbital electrons added since the last noble gas element (in column 18). This is in keeping with current interpretations of the periodic law which holds that the elements in a group have similar configurations of the outermost electron shells of their atoms. Since most chemical properties result from outer electron interactions, this tends to explain why elements in the same group exhibit similar physical and chemical properties. Unfortunately, the system fails for the elements in the first 3 periods (or rows; see below). For example, aluminum, in the column numbered 13, has only 3 s, p, and d orbital electrons. Nevertheless, the American Chemical Society has adopted the new IUPAC system.
The horizontal rows of the table are called periods. The elements of a period are characterized by the fact that they have the same number of electron shells; the number of electrons in these shells, which equals the element's atomic number, increases from left to right within each period. In each period the lighter metals appear on the left, the heavier metals in the center, and the nonmetals on the right. Elements on the borderline between metals and nonmetals are called metalloids.
Group 1 (with one valence electron) and Group 2 (with two valence electrons) are called the alkali metals and the alkaline-earth metals, respectively. Two series of elements branch off from Group 3, which contains the transition elements, or transition metals; elements 57 to 71 are called the lanthanide series, or rare earths, and elements 89 to 103 are called the actinide series, or radioactive rare earths; a third set, the superactinide series (elements 122-153), is predicted to fall outside the main body of the table, but none of these has yet been synthesized or isolated. The nonmetals in Group 17 (with seven valence electrons) are called the halogens. The elements grouped in the final column (Group 18) have no valence electrons and are called the inert gases, or noble gases, because they react chemically only with extreme difficulty.
In a relatively simple type of periodic table, each position gives the name and chemical symbol for the element assigned to that position; its atomic number; its atomic weight (the weighted average of the masses of its stable isotopes, based on a scale in which carbon-12 has a mass of 12); and its electron configuration, i.e., the distribution of its electrons by shells. The only exceptions are the positions of elements 103 through 118; complete information on these elements has not been compiled. Larger and more complicated periodic tables may also include the following information for each element: atomic diameter or radius; common valence numbers or oxidation states; melting point; boiling point; density; specific heat; Young's modulus; the quantum states of its valence electrons; type of crystal form; stable and radioactive isotopes; and type of magnetism exhibited by the element (paramagnetism or diamagnetism).
Organized array of all the chemical elements in approximately increasing order of their atomic weight. The elements show a periodic recurrence of certain properties, first discovered in 1869 by Dmitry I. Mendeleyev. Those in the same column (group) of the table as usually arranged have similar properties. In the 20th century, when the structure of atoms was understood, the table was seen to precisely reflect increasing order of atomic number. Members of the same group in the table have the same number of electrons in the outermost shells of their atoms and form bonds of the same type, usually with the same valence; the noble gases, with full outer shells, generally do not form bonds. The periodic table has thus greatly deepened understanding of bonding and chemical behaviour. It also allowed the prediction of new elements, many of which were later discovered or synthesized. For an illustration of the periodic table, see chemical element.
Some versions of the table show a dark stair-step line along the metalloids. Metals are to the left of the line and non-metals to the right.
Arrangement
The layout of the periodic table demonstrates recurring ("periodic") chemical properties. Elements are listed in order of increasing atomic number (i.e. the number of protons in the atomic nucleus). Rows are arranged so that elements with similar properties fall into the same vertical columns ( "groups" ). According to quantum mechanical theories of electron configuration within atoms, each horizontal row ( "period" ) in the table corresponded to the filling of a quantum shell of electrons. There are progressively longer periods further down the table, grouping the elements into s- , p- , d- and f-blocks to reflect their electron configuration.
In printed tables, each element is usually listed with its element symbol and atomic number; many versions of the table also list the element's atomic mass and other information, such as its abbreviated electron configuration, electronegativity and most common valence numbers.
As of 2006, the table contains 117 chemical elements whose discoveries have been confirmed. Ninety-four are found naturally on Earth, and the rest are synthetic elements that have been produced artificially in particle accelerators. Elements 43 (technetium), 61 (promethium), 93 (neptunium) and 94 (plutonium) have no stable isotopes and were first discovered synthetically; however, they were later discovered in trace amounts on earth as products of natural radioactive decay processes.
Periodicity of chemical properties
The main value of the periodic table is the ability to predict the chemical properties of an element based on its location on the table. It should be noted that the properties vary differently when moving vertically along the columns of the table than when moving horizontally along the rows.
Groups and periods
A group is a vertical column in the periodic table of the elements.
Groups are considered the most important method of classifying the elements. In some groups, the elements have very similar properties and exhibit a clear trend in properties down the group — these groups tend to be given trivial (unsystematic) names, e.g. the alkali metals, alkaline earth metals, halogens and noble gases. Some other groups in the periodic table display fewer similarities and/or vertical trends (for example Groups 14 and 15), and these have no trivial names and are referred to simply by their group numbers.
A period is a horizontal row in the periodic table of the elements.
Although groups are the most common way of classifying elements, there are some regions of the periodic table where the horizontal trends and similarities in properties are more significant than vertical group trends. This can be true in the d-block (or " transition metals"), and especially for the f-block, where the lanthanides and actinides form two substantial horizontal series of elements.
Periodic trends of groups
Modern quantum mechanical theories of atomic structure explain group trends by proposing that elements within the same group have the same electron configurations in their valence shell, which is the most important factor in accounting for their similar properties. Elements in the same group also show patterns in their atomic radius, ionization energy, and electronegativity. From top to bottom in a group, the atomic radii of the elements increase. Since there are more filled energy levels, valence electrons are found farther from the nucleus. From the top, each successive element has a lower ionization energy because it is easier to remove an electron since the atoms are less tightly bound. Similarly, a group will also see a top to bottom decrease in electronegativity due to an increasing distance between valence electrons and the nucleus.
Periodic trends of periods
Elements in the same period show trends in atomic radius, ionization energy, electron affinity, and electronegativity. Moving left to right across a period, atomic radius usually decreases. This occurs because each successive element has an added proton and electron which causes the electron to be drawn closer to the nucleus. This decrease in atomic radius also causes the ionization energy to increase when moving from left to right across a period. The more tightly bound an element is, the more energy is required to remove an electron. Similarly, electronegativity will increase in the same manner as ionization energy because of the amount of pull that is exerted on the electrons by the nucleus. Electron affinity also shows a slight trend across a period. Metals (left side of a period) generally have a lower electron affinity than nonmetals (right side of a period) with the exception of the noble gases.
Examples
Noble gases
All the elements of Group 18, the noble gases, have full valence shells. This means they do not need to react with other elements to attain a full shell, and are therefore much less reactive than other groups. Neon followed by helium are the most inert elements among noble gases, since reactivity, in this group, increases with the periods: it is possible to make heavy noble gases react since they have much larger electron shells. However, their reactivity remains very low in absolute terms.
Halogens
In Group 17, known as the halogens, elements are missing just one electron each to fill their shells. Therefore, in chemical reactions they tend to acquire electrons (the tendency to acquire electrons is called electronegativity). This property is most evident for fluorine (the most electronegative element of the whole table), and it diminishes with increasing period.
As a result, all halogens form acids with hydrogen, such as hydrofluoric acid, hydrochloric acid, hydrobromic acid and hydroiodic acid, all in the form HX . Their acidity increases with higher period, for example, with regard to iodine and fluorine, since a large I − ion is more stable in solution than a small F − , there is less volume in which to disperse the charge.
Transition metals
For the transition metals (Groups 3 to 12), horizontal trends across periods are often important as well as vertical trends down groups; the differences between groups adjacent are usually not dramatic. Transition metal reactions often involve coordinated species.
Lanthanides and actinides
The chemical properties of the lanthanides (elements 57–71) and the actinides (elements 89–103) are even more similar to each other than the transition metals, and separating a mixture of these can be very difficult. This is important in the chemical purification of uranium concerning nuclear power.
Structure of the periodic table
The primary determinant of an element's chemical properties is its electron configuration, particularly the valence shell electrons. For instance, any atoms with four valence electrons occupying p orbitals will exhibit some similarity. The type of orbital in which the atom's outermost electrons reside determines the "block" to which it belongs. The number of valence shell electrons determines the family, or group, to which the element belongs.
The total number of electron shells an atom has determines the period to which it belongs. Each shell is divided into different subshells, which as atomic number increases are filled in roughly this order (the Aufbau principle):
Note that as atomic number (i.e. charge on the atomic nucleus) increases, this leads to greater spin-orbit coupling between the nucleus and the electrons, reducing the validity of the quantum mechanical orbital approximation model, which considers each atomic orbital as a separate entity.
Because of the importance of the outermost shell, the different regions of the periodic table are sometimes referred to as periodic table blocks, named according to the sub-shell in which the "last" electron resides, e.g. the s -block, the p -block, the d -block, etc.
The elements Ununbium, ununtrium, ununquadium, etc. are elements that have been discovered, but so far have not been named.
In Ancient Greece, the influential Greek philosopher Aristotle proposed that there were four main elements: air, fire, earth and water. All of these elements could be reacted to create another one; e.g. , earth and fire combined to form lava. However, this theory was dismissed when the real chemical elements started being discovered. Scientists needed an easily accessible, well organized database with which information about the elements could be recorded and accessed. This was to be known as the periodic table.
The original table was created before the discovery of subatomic particles or the formulation of current quantum mechanical theories of atomic structure. If one orders the elements by atomic mass, and then plots certain other properties against atomic mass, one sees an undulation or periodicity to these properties as a function of atomic mass. The first to recognize these regularities was the German chemist Johann Wolfgang Döbereiner who, in 1829, noticed a number of triads of similar elements.
In 1829 Döbereiner proposed the Law of Triads: The middle element in the triad had atomic weight that was the average of the other two members. The densities of some triads followed a similar pattern. Soon other scientists found chemical relationships extended beyond triads. Fluorine was added to Cl/Br/I group; sulfur, oxygen, selenium and tellurium were grouped into a family; nitrogen, phosphorus, arsenic, antimony, and bismuth were classified as another group.
This was followed by the English chemist John Newlands, who noticed in 1865 that when placed in order of increasing atomic weight, elements of similar physical and chemical properties recurred at intervals of eight, which he likened to the octaves of music, though his law of octaves was ridiculed by his contemporaries. However, while successful for some elements, Newlands' law of octaves failed for two reasons:
It was not valid for elements that had atomic masses higher than Ca.
When further elements were discovered, such as the noble gases (He, Ne, Ar), they could not be accommodated in his table.
Finally, in 1869 the Russian chemistry professor Dmitri Ivanovich Mendeleev and four months later the German Julius Lothar Meyer independently developed the first periodic table, arranging the elements by mass. However, Mendeleev plotted a few elements out of strict mass sequence in order to make a better match to the properties of their neighbors in the table, corrected mistakes in the values of several atomic masses, and predicted the existence and properties of a few new elements in the empty cells of his table. Mendeleev was later vindicated by the discovery of the electronic structure of the elements in the late 19th and early 20th century.
Earlier attempts to list the elements to show the relationships between them (for example by Newlands) had usually involved putting them in order of atomic mass. Mendeleev's key insight in devising the periodic table was to lay out the elements to illustrate recurring ("periodic") chemical properties (even if this meant some of them were not in mass order), and to leave gaps for "missing" elements. Mendeleev used his table to predict the properties of these "missing elements", and many of them were indeed discovered and fit the predictions well.
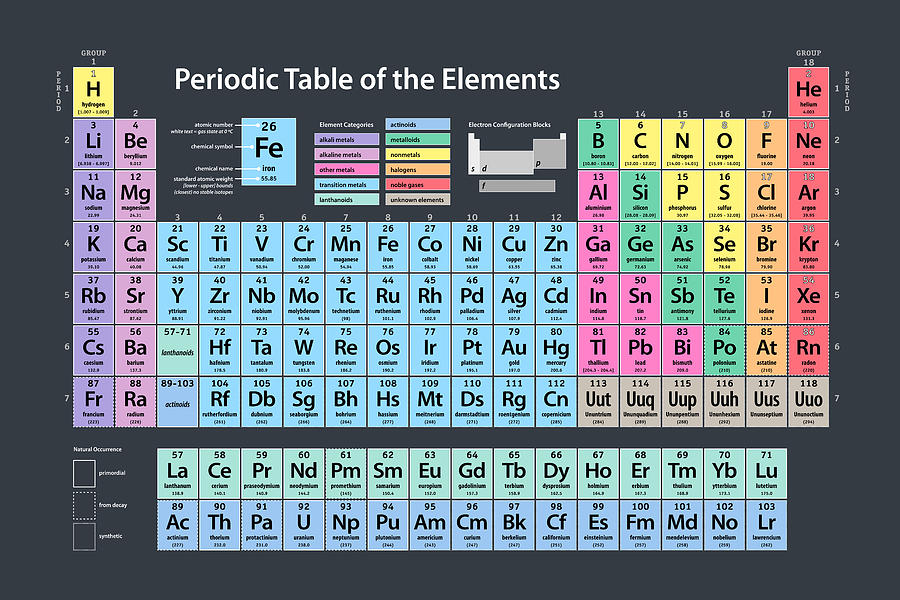

The pulchritudinous Periodic Table, created by Dimitri Mendeleev. Scroll down for a voluminated picture.